CDMO Services for Research and
Transfer Through CGMP Manufacturing
Large Molecule Expertise. Ingenuity You Can Count On.
20+ Years of CDMO Experience | 320+ Clinical Trials | 3+ Commercial Products
Welcome to Working with Cytovance Biologics CDMO
Our history of Ingenuity and Innovation is at your service.
Over two decades of collaborative scientific experience and technological manufacturing insight have empowered us to establish a unique Culture of Ingenuity that is a perfect match for your one-of-a-kind molecule. We leverage platform and custom manufacturing approaches in microbial and mammalian expression systems with a solutions-oriented mindset that propels your asset to its next milestone. As your trusted CDMO partner, responsible in part for your success, we promise to always be Responsive, Reliable, and Resourceful.
Complex Molecules. Creative CDMO Solutions.
Where can we help you on the path to commercialization?
Large molecules are our core expertise. Our team provides world-class strain and process development, manufacturing, analytical, and regulatory capabilities that enable our clients to produce antibodies, fusion proteins, enzymes, vaccines, recombinant proteins and others for critical medical needs.
Mammalian
Cell line development to expression of complex recombinant proteins in mammalian cell culture.
Pre-Commercial and Commercial Services
Manufacturing process characterization, validation, and support for regulatory success and beyond.
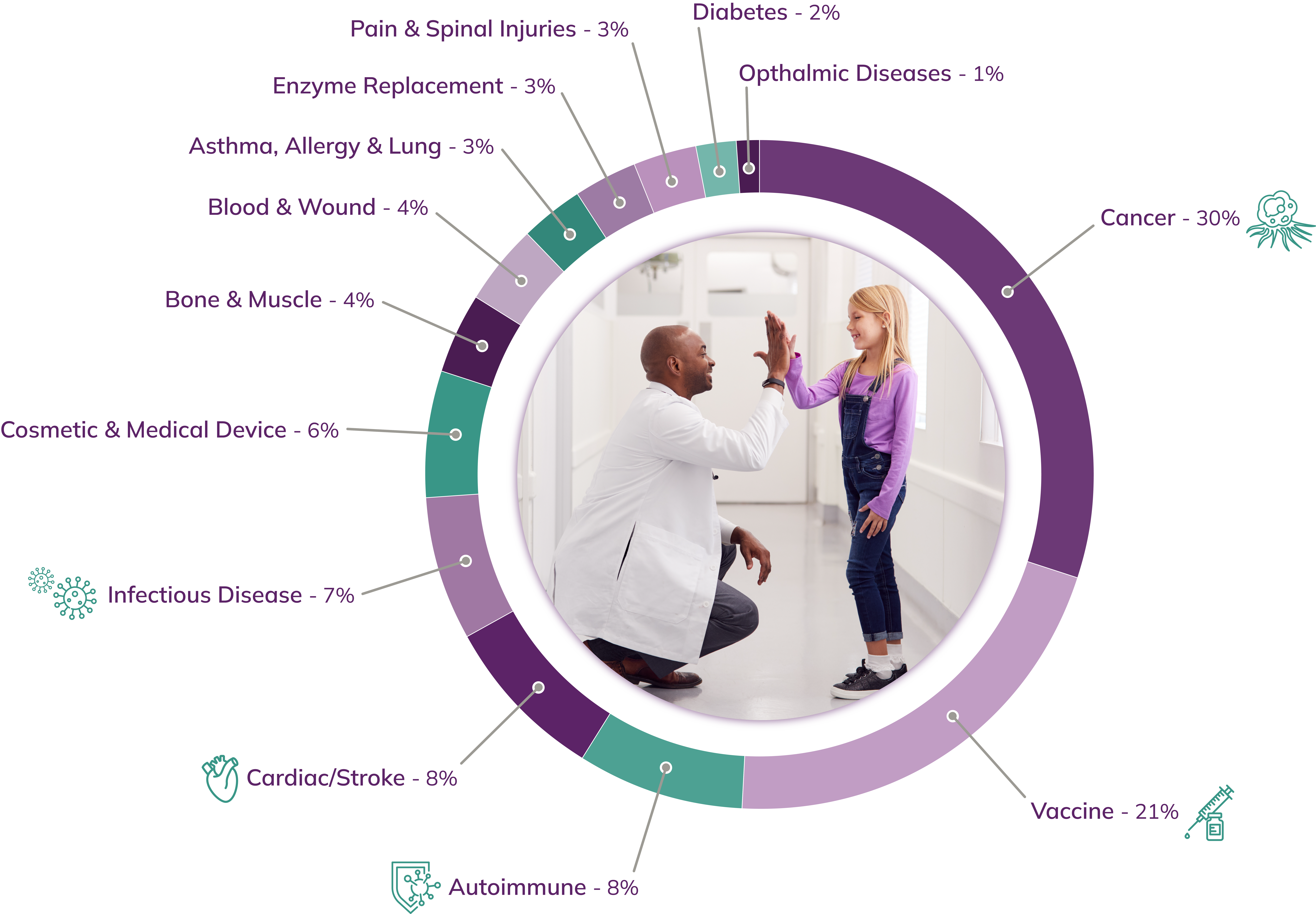
Manufacturing Solutions Across Indications
Cytovance’s operational excellence, seamless integration of quality controls, and dedication to regulatory compliance efficiently deliver success to our customers and safe, effective treatments to patients. We are proud to have supported hundreds of clinical trials and commercial products. How can our team support your program?
Biologics CDMO Industry News and Insights
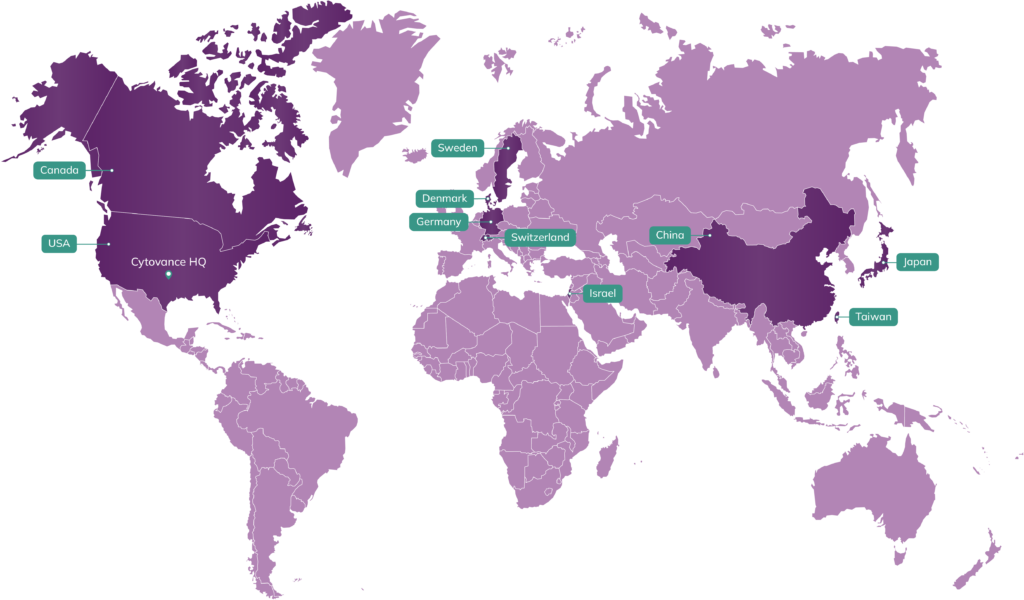
Oklahoma Roots, Global Reach
With a strategic location in the American Midwest, Cytovance is experienced in servicing global biopharma with resourceful DNA sequence to cGMP manufacturing services, responsive program management, and reliable logistics. Our 200+ international customers are predominantly based out of North America, Europe, and the APAC region.
The Company We Keep
We are revolutionizing the industry through best-of-breed partnerships with one goal in mind: delivering the best service for you.







Never miss a thing – sign up for our periodic LinkedIn newsletter.
Making a Difference in America’s Heartland
Committed to Growing Biotech in Oklahoma City
We’re proud to be the largest biotechnology company in Oklahoma! Our strategic Midwest location allows us to pass on the benefits of centralized logistics, smart cost structures, and ever-growing cGMP capacity to our industry clients.
At the center of the Oklahoma City Innovation District, you’ll find our campus headquarters and the heart of a flourishing $7 billion biotechnology community committed to collaboration, innovation, and economic growth. We have partnered with the University of Oklahoma and other institutions to foster strong educational initiatives and a biomanufacturing workforce training program. Together, we’re inspiring students, scientists, and entrepreneurs while enriching the future of biotechnology and biomanufacturing in America’s Heartland.

Contact Us
Meet Team Cytovance in 2026!
-
– BIO Investment and Growth Summit: March 2-3, Miami Beach, FL
-
– OPCU Large Molecule: March 24, Virtual
-
– World Vaccine Congress: March 30-April 2, Washington D.C. | Booth 536
– PEGS: May 11-15, Boston, MA -
– TIDES USA Oligonucleotide & Peptide Therapeutics: May 11-14, Boston, MA
-
– BIO International Convention: June 22-25, San Diego, CA | Booth 4445
-
– BioProcess International East: September 22-25, Boston, MA
-
– CPHI Europe: October 6-8, Milan, Italy
-
– CDMO Live Americas: October 19-21, Boston, MA
- – OPCU Large Molecule: October 27, Virtual
