Mammalian Biologics CDMO Services
Biomanufacturing Support for Your Mammalian-Derived Products
Mammalian-derived recombinant protein production represents a core capability of Cytovance Biologics. Our extensive experience, combined with our deep technical expertise, has enabled the progression of multiple complex proteins from early phase process development through scaled-up cGMP manufacturing for clinical evaluation. With a broad range of production scales up to 2000L and integrated development capabilities, we offer you both fully custom and platformed approaches to your mammalian biologic’s success.
We provide flexible end-to-end development of all ranges of complex molecules:
• Antibodies
• Fusion Proteins
• Bispecific
• TriKE
• Cytokines
• Enzymes

Keep Your Molecule Moving Forward
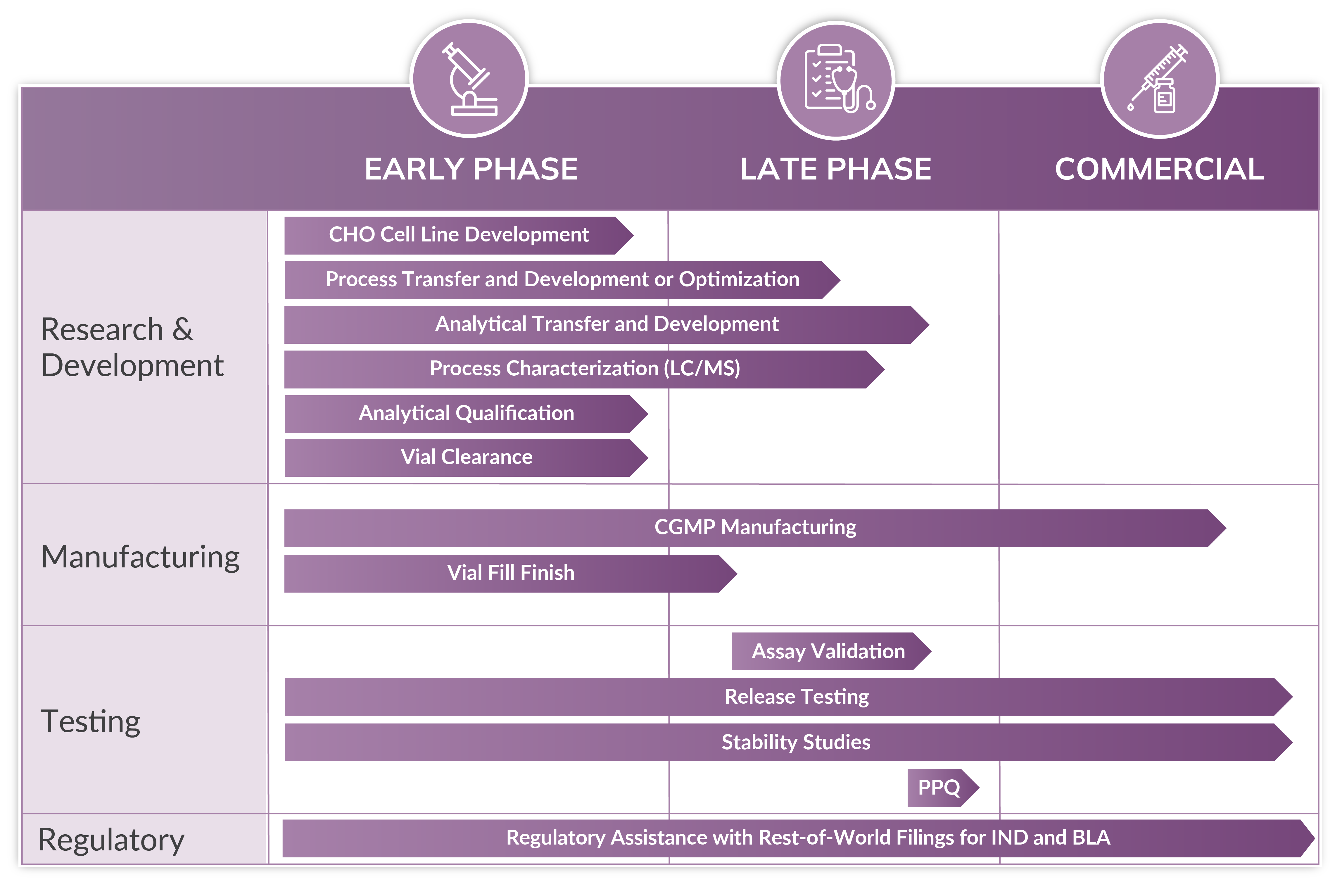
Cell Line Development
Complete mammalian clone selection, clonal stability, and banking services tailored to your requirements.
Mammalian Process Development
Take your productivity to the next level with our process development experts.
Mammalian Analytical Development
Robust analytical method development or transfer personalized for your mammalian-derived biologics.
Mammalian Manufacturing
Benefit from our full-service cGMP facility from cell banks to production with up to 2,000L capacity.
The Power of Partnership
“We recently celebrated successful scale-up and GMP release of the first 1000L DS. We were very lucky to succeed on our first go as a byproduct of Team Cytovance’s hard work.”
— Scientist and Program Lead, Medium Biotech
Contact Us
Meet Team Cytovance in 2026!
-
– BIO Investment and Growth Summit: March 2-3, Miami Beach, FL
-
– OPCU Large Molecule: March 24, Virtual
-
– World Vaccine Congress: March 30-April 2, Washington D.C. | Booth 536
– PEGS: May 11-15, Boston, MA -
– TIDES USA Oligonucleotide & Peptide Therapeutics: May 11-14, Boston, MA
-
– BIO International Convention: June 22-25, San Diego, CA | Booth 4445
-
– BioProcess International East: September 22-25, Boston, MA
-
– CPHI Europe: October 6-8, Milan, Italy
-
– CDMO Live Americas: October 19-21, Boston, MA
- – OPCU Large Molecule: October 27, Virtual
