Microbial CDMO Services
Biomanufacturing Support for Your Microbial Expressed Products
With over 84 proteins successfully expressed in the past decade, we are microbial CDMO experts! When you partner with us, you have our deep technical expertise in bacteria and yeast batch and fed batch-based fermentation processes to help guide your microbial-derived biologic from end to end. Whether from early-phase process development and optimization for scale-up cGMP manufacturing or late-stage manufacturing, we offer the flexibility and ingenuity to build on the work that your team has already accomplished. Our robust manufacturing capacity in the United States and impressive titers, combined with integrated quality and regulatory oversight, give you the peace of mind needed to reliably advance your biologic through the product lifecycle.

Keep Your Molecule Moving Forward
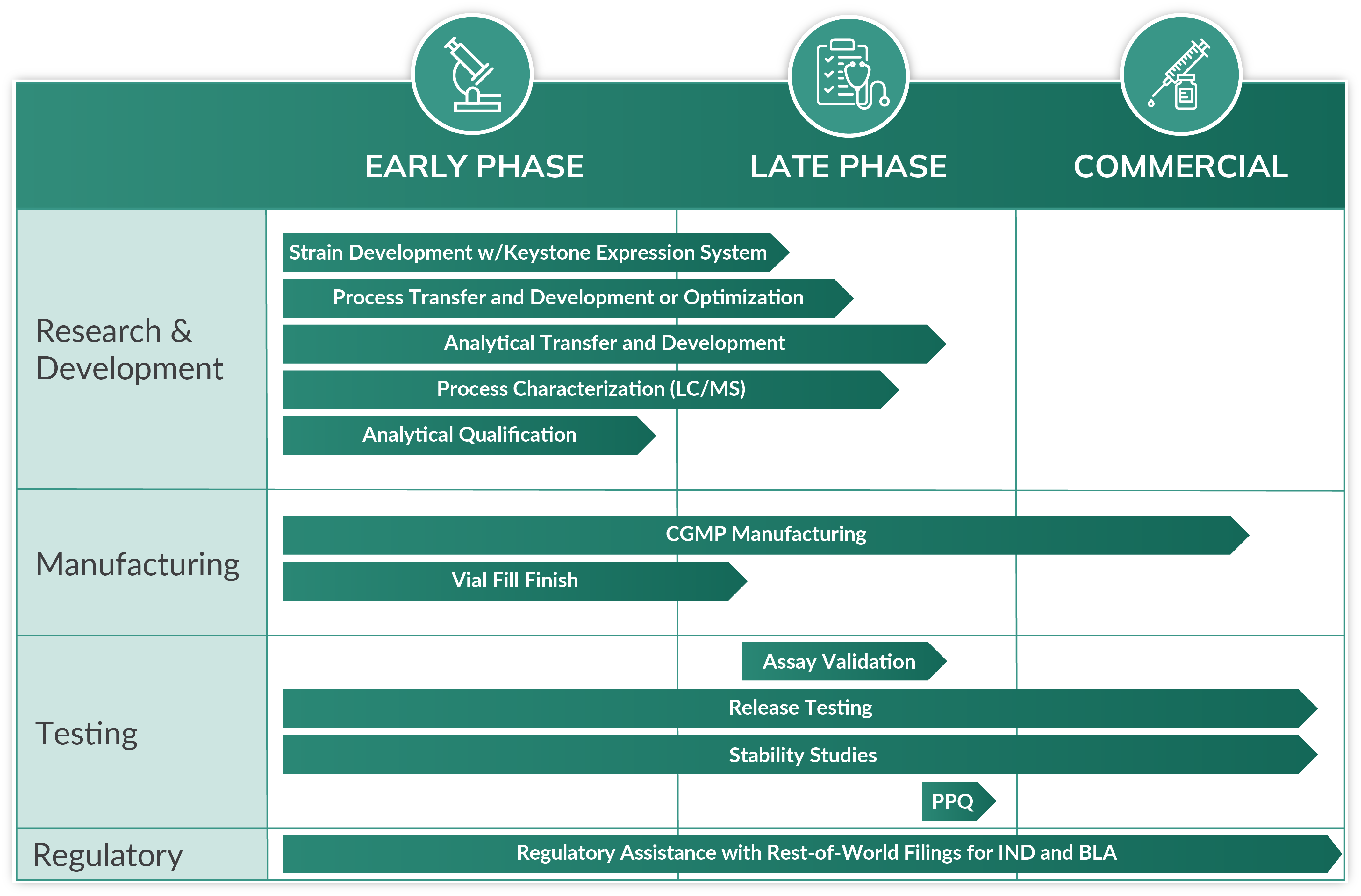
Microbial Analytical Development
Robust analytical development personalized for your drug substance or biologic.
Microbial Manufacturing
Benefit from our full-service cGMP facility with up to 1,000L microbial capacity.
Ingenuity, On Demand
Thought Leadership from Team Cytovance
A Strain Development Strategy to Enhance Screening, Process Development, and Scale-Up
A Brief History of Microbial Biomanufacturing
Downstream Process Development of a Complicated Fusion Protein
Key Considerations for Choosing the Right Microbial CDMO Partner
General Scale-Up Strategies: Commonalities Between Mammalian and Microbial Processes
Mammalian or Microbial? Considerations When Choosing an Expression System for Your Product
The Power of Partnership
“Our recent product release milestone would not have been achieved if it were not for the dedication, hard work, and collaboration from Team Cytovance.”
— Program Lead, F500 Pharma
Contact Us
Meet Team Cytovance in 2026!
-
– BIO Investment and Growth Summit: March 2-3, Miami Beach, FL
-
– OPCU Large Molecule: March 24, Virtual
-
– World Vaccine Congress: March 30-April 2, Washington D.C. | Booth 536
– PEGS: May 11-15, Boston, MA -
– TIDES USA Oligonucleotide & Peptide Therapeutics: May 11-14, Boston, MA
-
– BIO International Convention: June 22-25, San Diego, CA | Booth 4445
-
– BioProcess International East: September 22-25, Boston, MA
-
– CPHI Europe: October 6-8, Milan, Italy
-
– CDMO Live Americas: October 19-21, Boston, MA
- – OPCU Large Molecule: October 27, Virtual
