Who We Are
A Biologics CDMO Driven By Your Success
Cytovance Biologics is a leader in the development and manufacturing of clinical and commercial large molecules from mammalian cell culture and microbial fermentation.
Cytovance delivers scientific rigor, a solutions-oriented mindset, and ingenuity end-to-end. When combined with our comprehensive service portfolio of strain and cell line development process development, analytical capabilities, cell banking, scale-up, cGMP manufacturing, program management, and lifecycle support, we are able to support products throughout the journey from R&D to regulatory approval and commercial production. Our team’s approach and breadth of knowledge have led to our high success rate and multiple client products on the market, actively making the world a healthier place.

Our Culture of Ingenuity
Inspired Minds Developing Strong Solutions
Every day, our Responsive nature, Reliable processes, and Resourceful approach combine to create a Culture of Ingenuity that delivers complex large molecule development and manufacturing success to our partner. We hire scientists, program managers, and logistics staff with solution-oriented mindsets and out-of-the-box creativity. Then, we empower these great minds to not only embrace their individuality and passion but to clearly see the connection of their biologics CDMO work to enabling better outcomes for patients around the world. When you partner with Cytovance, we are with you every step of the way to ensure the success of your molecule because we believe and understand the impact it can have in treating unmet medical needs.
Resourceful
Team Cytovance leverages early-stage analytical insight and common stock options to proactively overcome production obstacles.
Responsive
When unexpected challenges arise, or a partner has complex product needs, we fit our processes to them and look for solutions.

Reliable
Our high success rate means our partners can depend on our technical knowledge to deliver complex molecules to their next milestone on the path to market timeline.
Our Vision
We believe in a healthier world where every biologic’s path to commercialization is advanced by a development and manufacturing partner that delivers ingenuity end-to-end.
Our Mission
As your trusted biologics CDMO partner, our culture of ingenuity enables the development and manufacture of therapeutics, from clinical to commercial. We are responsive to your processes and needs, reliable in our technical expertise, and resourceful in our approach to your success.
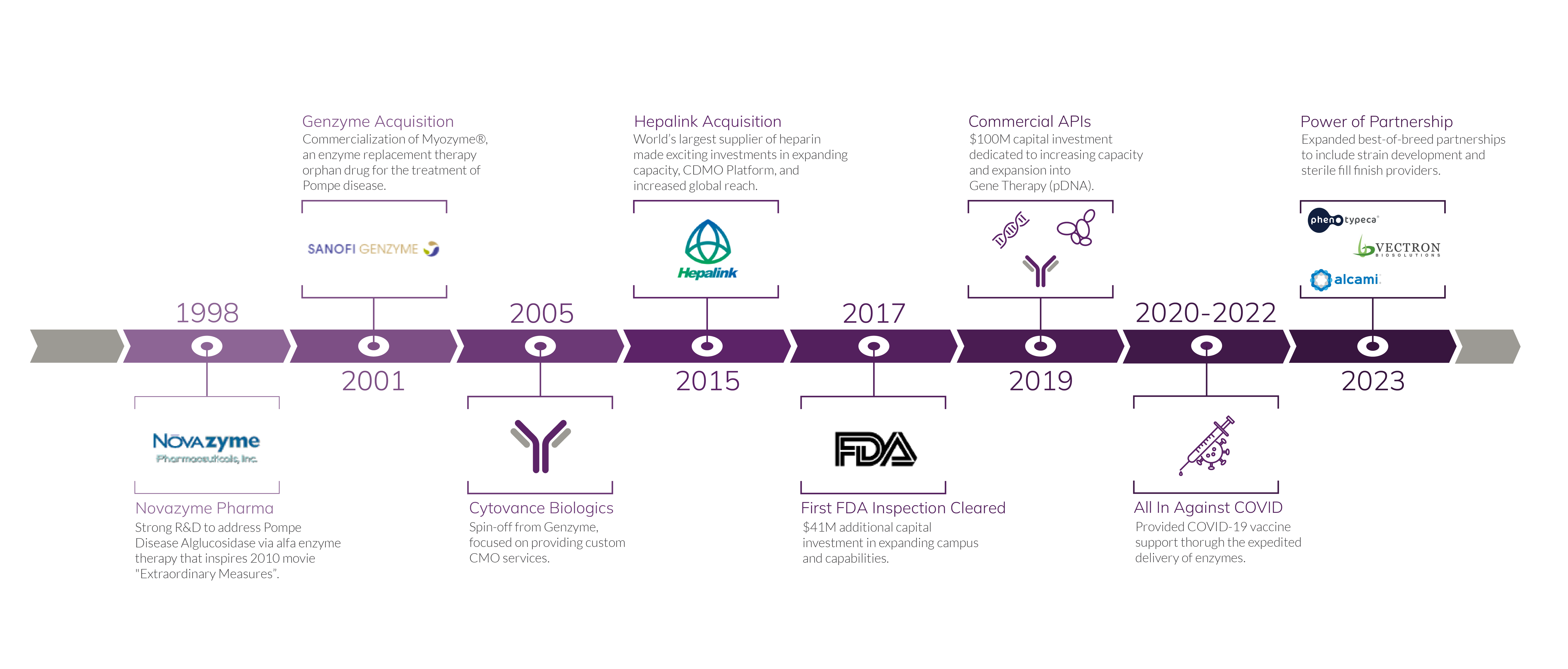
A History of Innovation and Ingenuity
Decades of Scientific and Manufacturing Expertise
Contact Us
Meet Team Cytovance in 2026!
-
– BIO Investment and Growth Summit: March 2-3, Miami Beach, FL
-
– OPCU Large Molecule: March 24, Virtual
-
– World Vaccine Congress: March 30-April 2, Washington D.C. | Booth 536
– PEGS: May 11-15, Boston, MA -
– TIDES USA Oligonucleotide & Peptide Therapeutics: May 11-14, Boston, MA
-
– BIO International Convention: June 22-25, San Diego, CA | Booth 4445
-
– BioProcess International East: September 22-25, Boston, MA
-
– CPHI Europe: October 6-8, Milan, Italy
-
– CDMO Live Americas: October 19-21, Boston, MA
- – OPCU Large Molecule: October 27, Virtual
